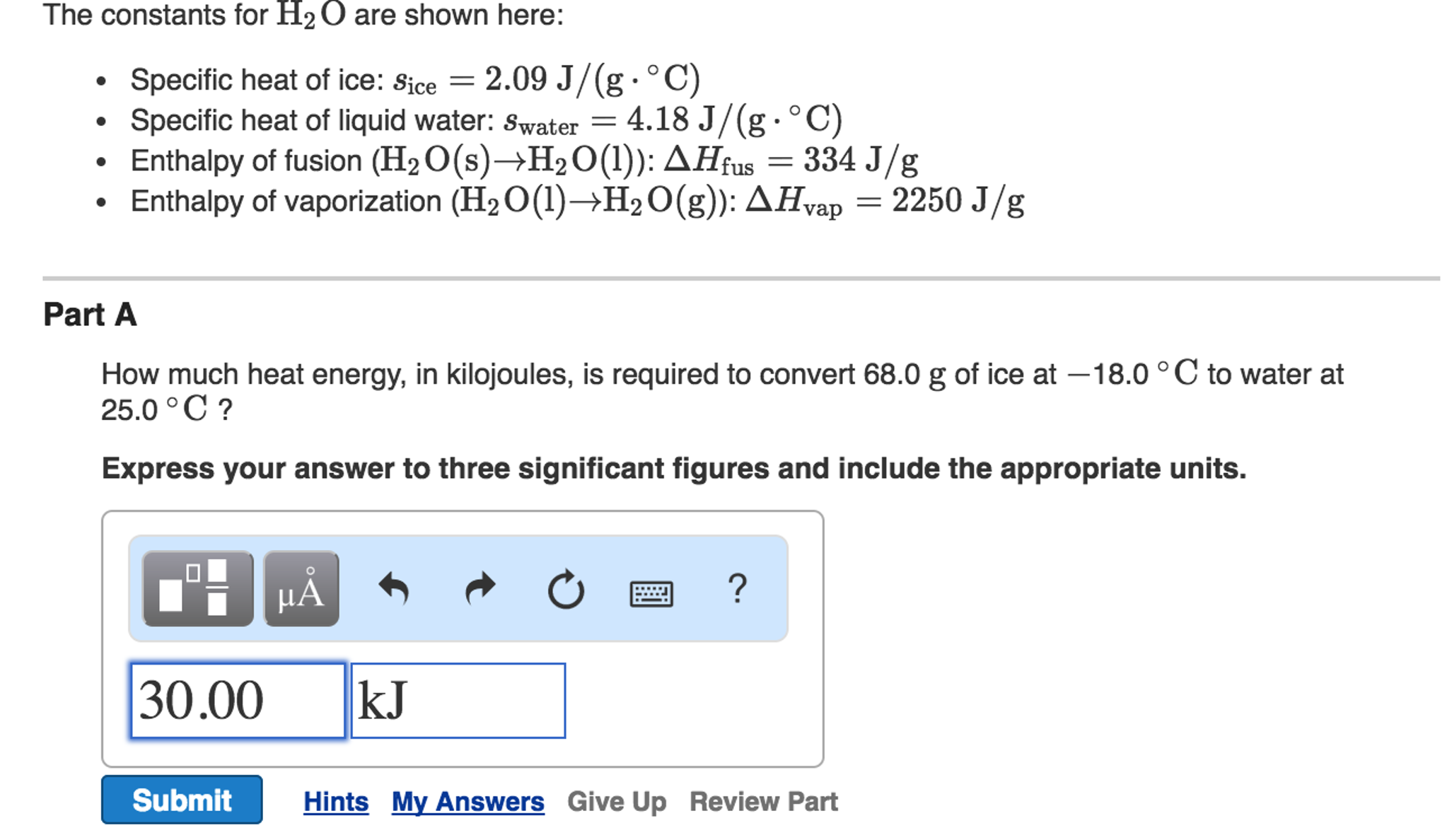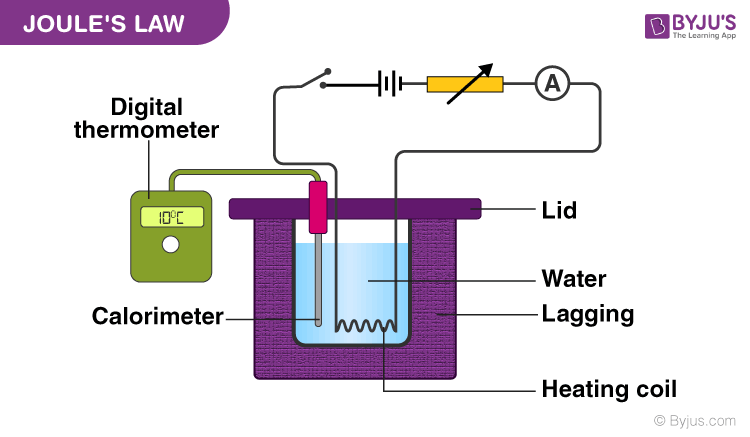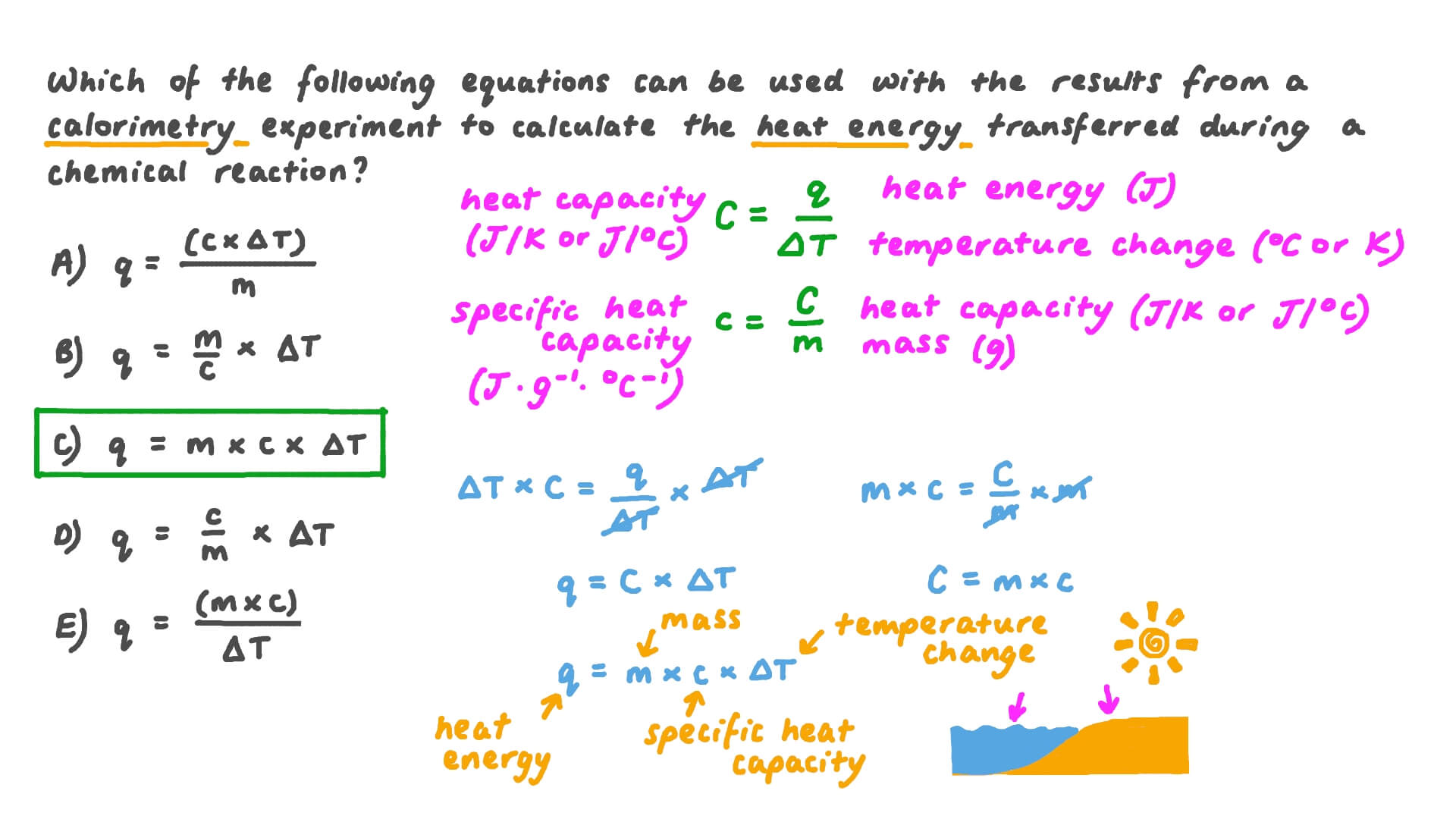
Question Video: Determining the Correct Formula to Use in Order to Calculate the Heat Transferred in a Calorimetry Experiment | Nagwa

Kirkebøker: Ministerialprotokoller, klokkerbøker og fødselsregistre - Nordland, SAT/A-1459/872/L1049: Klokkerbok nr. 872C05, 1920-1938, s. 164 - Skanna arkiver - Arkivverket

108 (Fortegnelse over Filologer, Realister og Mineraloger fra Norges Universitet 1813-1884 : samt Kalender for de høiere norske Skoler)

When 1 g of water change from liquid to vapour phase at constant pressure of 1 atmosphere, the volume increases from 1 C to 1671 C . The heat of vaporisation at

CHEMISTRY September 21, Warm Up What is the difference between heat and temperature? Which bench will you sit on when exposed to 120 o F sunlight. - ppt download

Kinetics of Methanol Synthesis from Carbon Dioxide Hydrogenation over Copper–Zinc Oxide Catalysts | Industrial & Engineering Chemistry Research
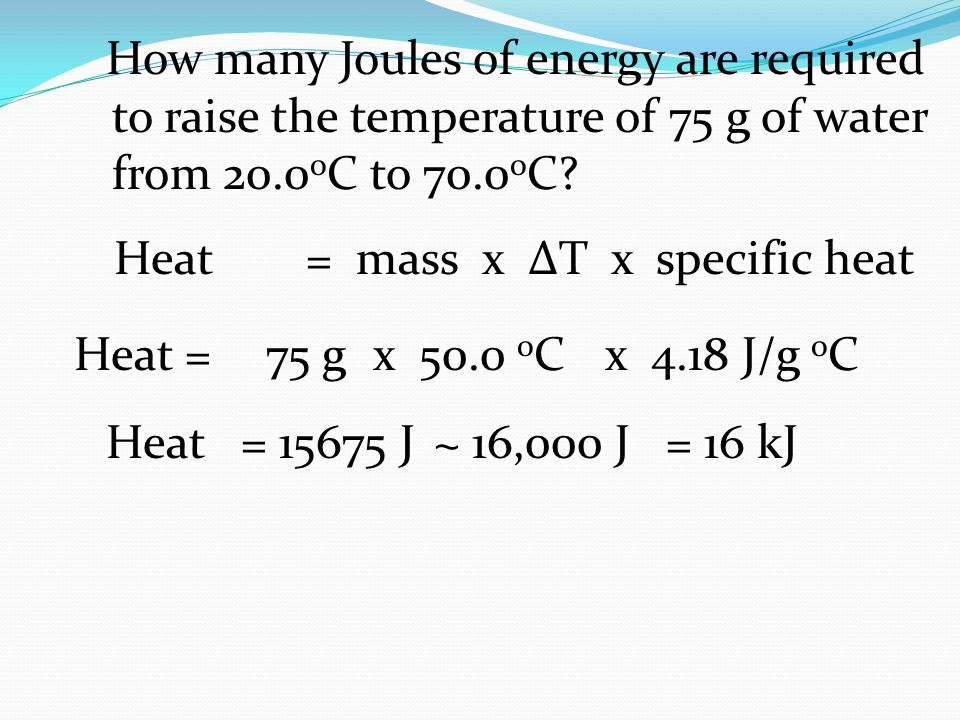
How many Joules of energy are required to raise the temperature of 75 g of water from 20.0 o C to 70.0 o C? Heat =75 g x 50.0 o C x

Specific Heat The amount of heat energy it takes to raise the temperature of 1 gram of a substance by 1 degree Celsius Specific Heat of water = 4.18 Joules/g0C. - ppt download


:max_bytes(150000):strip_icc()/Five-C-479128409bd247e596d17bf7fb363b08.jpg)